Hospital and Medical Facility Cleaning Standards and Compliance in Australia: Ensuring Infection Control and Regulatory Adherence
Hospital and medical facility cleaning standards in Australia set the baseline for infection prevention, patient safety and regulatory compliance across public and private healthcare settings. This article explains the standards that drive environmental hygiene, the mechanisms by which effective cleaning reduces healthcare-associated infections (HAIs), and practical protocols for implementing risk-based programmes in clinics, theatres and critical care units. Readers will learn how national frameworks such as the NSQHS Standards intersect with Queensland Health guidance, how Therapeutic Goods Administration (TGA) considerations determine disinfectant selection, and what a defensible audit trail looks like for facility managers. The guide also outlines specific operational steps — from high-touch surface disinfection to terminal cleaning of operating theatres — and includes practical tables and checklists to support compliance and quality assurance. Throughout, the content integrates local context for Sunshine Coast and Brisbane medical centres and explains how a compliant commercial cleaning partner can support documented infection control programmes.
What Are the Essential Healthcare Cleaning Standards in Australia?
Essential healthcare cleaning standards in Australia are a combination of national frameworks and state guidance that require risk-based environmental cleaning, use of approved hospital-grade disinfectants, documented cleaning schedules and audit evidence to demonstrate effectiveness. These standards aim to reduce pathogen transmission, protect vulnerable patients and provide traceable compliance records for regulators. Practical facility programmes map national NSQHS expectations to local clinic workflows and ensure staff competency, written procedures and product safety data are available on site. Understanding these overlapping requirements allows managers to prioritise interventions that both reduce HAIs and satisfy accreditation inspections.
This table summarises the main authorities and their primary cleaning requirements to clarify areas of overlap and responsibility for facility managers.
| Authority | Primary Cleaning Requirement | Key Practical Action |
|---|---|---|
| NSQHS Standards (Standard 3) | Risk-based environmental cleaning; documented processes | Develop cleaning matrix, audits, staff training |
| Australian Guidelines for the Prevention and Control of Infection in Healthcare (2019) | Evidence-based procedures for routine and specialist areas | Adopt standardised cleaning and disinfection methods |
| Therapeutic Goods Administration (TGA) | Use of listed/ARTG products for clinical uses where required | Select TGA-listed disinfectants and verify labels/MSDS |
| Queensland Health guidance | Local reporting, outbreak response protocols and clinic-specific requirements | Align local SOPs and reporting lines with state guidance |
This comparison highlights that the common practice points are a risk-based cleaning matrix, use of appropriate disinfectants, documented audits and staff competency records — all of which create a defensible compliance position.
How Do NSQHS Standards Guide Medical Facility Cleaning Compliance?
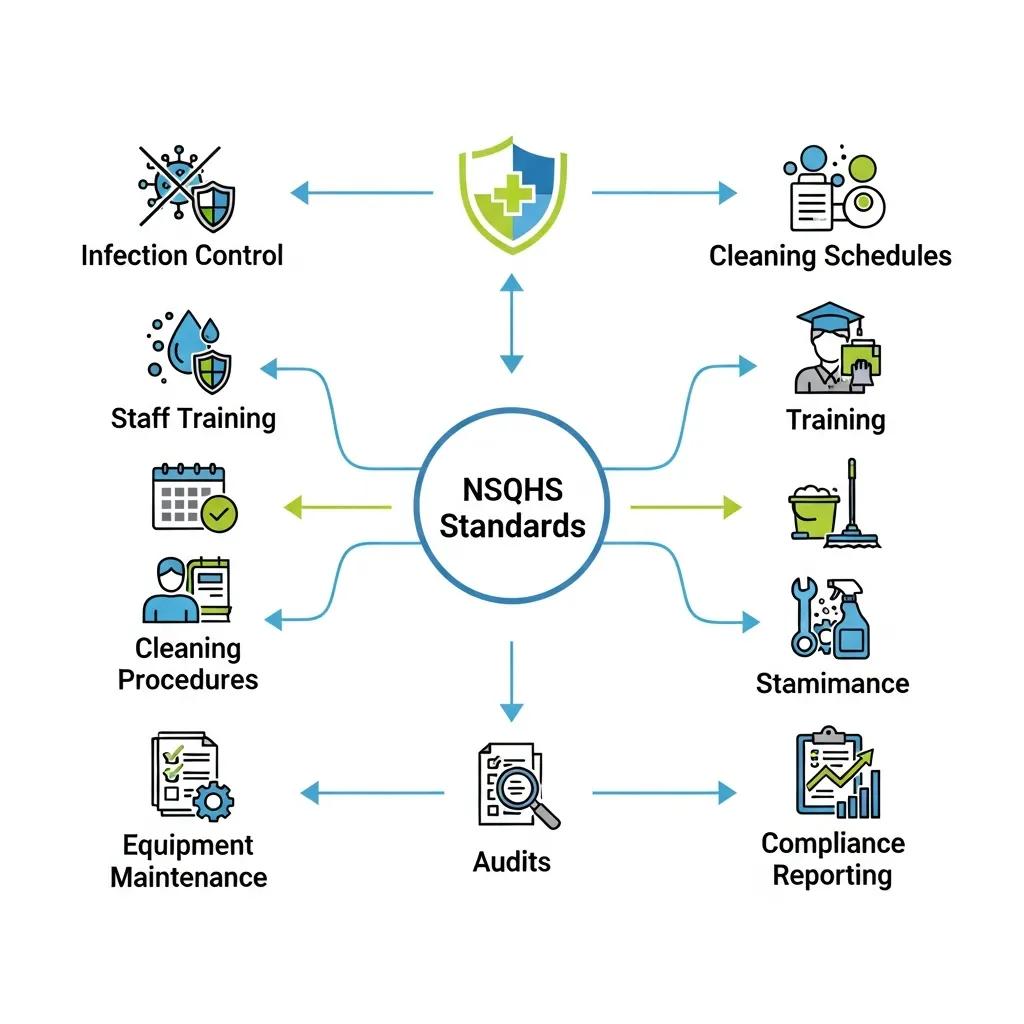
NSQHS Standard 3 focuses on minimising infection risks through systematic environmental hygiene, policies and measurable performance indicators, and it requires facilities to demonstrate governance and operational control of cleaning programmes. Facilities meet this standard by documenting cleaning policies, maintaining a risk-based schedule that differentiates high and low-risk zones, and performing regular audits with corrective-action records that show continuous improvement. Practical compliance steps include mapping clinical pathways to cleaning frequency, maintaining product MSDS and ensuring staff training records are available for inspection. Demonstrable examples — such as between-case cleaning logs for procedure rooms and routine ATP testing trends — strengthen a facility’s evidence for NSQHS conformity.
These operational controls naturally feed into state-level requirements, which add reporting and local protocol specificity where clinics must adapt national expectations to their context.
What Queensland Health Guidelines Apply to Clinic Cleaning Protocols?
Queensland Health provides clinic-level guidance that complements national frameworks by emphasising local outbreak reporting, specific documentation for outpatient settings and alignment with public health notification processes. Clinics on the Sunshine Coast and Brisbane should therefore adopt national cleaning matrices while incorporating state reporting triggers and any mandated notification timelines for certain HAIs or incidents. Actionable steps for clinics include establishing local escalation pathways for suspected outbreaks, keeping state-specific records of cleaning audits and ensuring communication lines with facility infection control teams. By aligning national standards with Queensland Health expectations, clinics can ensure seamless regulatory compliance and timely responses to infection control events.
These state-specific details reinforce the need for consistent training and documentation that demonstrate both routine and exceptional cleaning actions to auditors.
How Does Infection Control Prevent Healthcare-Associated Infections?
Infection control prevents healthcare-associated infections by interrupting the chain of transmission through targeted environmental cleaning, hand hygiene facilitation and staff behaviours that reduce pathogen spread. The mechanism is straightforward: removing organic soil and decontaminating surfaces reduces microbial load, while trained staff and validated cleaning processes prevent recontamination and cross-transmission. Effective programmes measure outcomes with environmental monitoring and link those results to corrective actions so that risk is continually managed and reduced. Recent studies and guideline updates in the current timeframe support combined approaches — cleaning, disinfection, auditing and staff competency — to achieve measurable reductions in HAIs.
Below is a concise, actionable list of core infection control steps that facilities should prioritise; the list is designed for quick operational adoption.
- Clean first, then disinfect: remove organic matter before applying disinfectant to ensure efficacy.
- Focus on high-touch surfaces: schedule frequent targeted cleaning for items touched repeatedly.
- Use appropriate contact/dwell times: follow product label instructions for pathogen kill.
- Maintain PPE and hand-hygiene practices: protect staff and reduce cross-contamination.
- Monitor and audit regularly: use ATP or surface swabs and close corrective actions.
These steps form a practical backbone for clinical cleaning programmes and lead directly into specific protocols for routine actions, spill responses and complex-area cleaning.
What Are the Key Infection Control Protocols for Medical Facilities?

Key infection control protocols include a defined cleaning sequence (clean then disinfect), contact precautions for infectious patients, safe handling and disposal of clinical waste, and immediate containment and cleaning of spills or bodily fluid incidents. Each protocol is justified by its role in interrupting transmission: sequence prevents disinfectant inactivation by soils, contact precautions limit spread via staff and equipment, and clinical waste procedures avoid environmental contamination. Facilities should codify these protocols into SOPs, provide scenario-based training and keep incident logs to enable traceability. Implementing these protocols consistently reduces risk and supports a defensible infection control posture during audits.
Clear documentation of these protocols also facilitates staff understanding and helps auditors verify that planned actions match executed routines.
How Are High-Touch Surfaces Disinfected to Minimise Cross-Contamination?
High-touch surface disinfection targets items such as bed rails, door handles, light switches, tabletops and clinical equipment controls with a frequency and method proportional to their contamination risk. Best practice uses colour-coded microfibre systems to prevent cross-use, wipes or solutions with validated contact times, and routine verification such as ATP testing to confirm cleaning quality. Staff should follow a standardised approach: inspect, clean with detergent if soiled, apply disinfectant with correct contact time, and document completion. Training reinforces technique, and photographic or digital audit trails help demonstrate consistent execution.
Establishing these practices lowers environmental bioburden and integrates seamlessly into risk-based cleaning schedules for higher-acuity areas like theatres and ICUs.
Which TGA Approved Hospital-Grade Disinfectants Are Used in Healthcare Cleaning?
TGA approval or ARTG listing for disinfectants signals that products meet regulatory expectations for specific clinical uses, including validated active ingredients and manufacturer claims relevant to healthcare settings. Common hospital-grade product categories include chlorine-releasing agents, quaternary ammonium compounds, and hydrogen peroxide-based systems — each chosen for compatibility with surfaces, required spectrum of activity and safety profile. Facilities must verify product labels and MSDS for intended uses, contact times and compatibility with medical equipment, and ensure staff follow manufacturer instructions to achieve expected pathogen reduction. Proper product selection balances efficacy, material compatibility and workplace safety.
This table compares broad disinfectant categories, the TGA-related approval attributes to check, and typical use cases to assist procurement decisions.
| Product Category | TGA Approval Criteria to Verify | Typical Healthcare Use Case |
|---|---|---|
| Chlorine-releasing agents | Efficacy claims against viruses/bacteria; corrosion warnings | Terminal cleaning, hard-surface disinfection in high organic load areas |
| Quaternary ammonium compounds (quats) | Spectrum claims and compatibility data | Routine surface disinfection in wards and communal areas |
| Hydrogen peroxide systems | Stabilised formulations and contact-time data | Delicate equipment areas and rapid-action surface disinfection |
This comparison helps teams choose appropriate classes of disinfectants while ensuring compliance with TGA expectations and facility safety needs.
Divine Commercial Cleaning maintains a policy of specifying TGA-listed, hospital-grade disinfectants and offers sustainable product options where compatible with clinical requirements; clients may request product lists and MSDS for review as part of procurement and risk assessments. This practice supports both regulatory adherence and environmentally conscious cleaning programmes.
What Criteria Define TGA Approval for Medical Cleaning Products?
TGA approval (or an ARTG listing where applicable) assesses a product’s intended claims, active ingredients, concentration and evidence supporting efficacy for clinical uses, while manufacturers provide MSDS and usage instructions that end-users must follow. Facilities should confirm that the product’s claims match the intended use — for example, surface disinfection in a clinical setting — and review contact times, dilution requirements and material compatibility. Practical verification steps include checking the manufacturer’s technical data, retaining MSDS on site and documenting product selection rationale in procurement records. These steps ensure both regulatory alignment and operational safety.
Verifying these criteria also informs training content so staff understand why certain products are used in specific areas and how to apply them correctly.
How Do Hospital-Grade Disinfectants Ensure Effective Pathogen Elimination?
Hospital-grade disinfectants achieve pathogen elimination through active chemistries that disrupt microbial structures, provided they are applied correctly with adequate contact time and on surfaces free from interfering organic matter. Mechanisms vary by class — oxidising agents denature proteins, quats disrupt membranes, and chlorine compounds oxidise cellular components — but all require adherence to label instructions for concentration and dwell time. Facilities validate performance through monitoring methods such as ATP testing or microbiological swabs and by tracking cleaning audit results to confirm trends. Effective elimination is therefore a combination of product selection, correct application and objective verification.
Ongoing validation is important because real-world factors like organic load and surface type influence efficacy, which is why audit data must inform product use decisions.
What Is Risk-Based Cleaning and How Is It Applied in Medical Environments?
Risk-based cleaning is a systematic approach that classifies areas by patient vulnerability, procedure invasiveness and traffic to assign cleaning frequencies and procedures that proportionally reduce infection risk. The purpose is to deploy resources where they have greatest impact — for example, between-case cleaning in operating theatres and hourly high-touch cleaning in intensive care units — while maintaining safe baseline cleaning for administrative spaces. A risk matrix clarifies categories and recommended actions, enabling consistent scheduling, auditability and resource planning. Applying this model helps facilities meet NSQHS expectations for targeted infection prevention and demonstrates a reasoned compliance strategy to auditors.
The following risk matrix provides a practical reference for classifying areas and matching actions.
| Area Type | Risk Category | Recommended Frequency/Action |
|---|---|---|
| Operating theatre / ICU | High | Between-case cleaning, terminal cleaning after list, ATP verification |
| Procedure rooms / Treatment bays | Medium | Between-patient surface cleaning, daily deep clean |
| Waiting areas / Offices | Low | Daily routine cleaning, targeted high-touch cleaning |
How Are High, Medium, and Low-Risk Areas Identified and Cleaned?
High-risk areas are identified by patient vulnerability and procedure invasiveness (e.g., operating theatres, ICUs), medium-risk zones by direct patient contact but lower invasiveness (e.g., procedure rooms), and low-risk areas by limited clinical exposure (e.g., admin offices). Identification uses criteria such as aerosol-generating procedures, immunocompromised populations and traffic patterns to create a risk score for each space. Cleaning protocols then specify frequency, product class and validation method for each risk tier — high-risk areas require hospital-grade disinfectants and verification, while low-risk areas use standard detergents and routine checks. This structured approach ensures cleaning resources prioritise patient safety and regulatory expectations.
Applying a documented risk scoring process provides defensible rationale for cleaning schedules and supports transparent audit records.
What Cleaning Frequencies and Procedures Are Recommended for Operating Theatres and ICUs?
Operating theatres and ICUs demand the most stringent cleaning: between-case cleaning for surfaces touched during a procedure, daily deep cleaning of all horizontal and vertical surfaces, and terminal cleaning after lists or patient discharge that includes detailed checks and validation. Between-case cleaning focuses on high-touch items and immediate patient-adjacent surfaces, while terminal cleaning includes mopping, patient transport route cleaning and ATP or swab verification where indicated. Schedules should specify who performs each task, required contact times for disinfectants and documentation sign-off to provide an audit trail. Regular competency assessments and incident-triggered deep cleans complete the programme for these very high-risk zones.
Consistent application of these procedures and evidence of validation are essential to demonstrate infection control effectiveness to accreditation bodies.
What Specialised Cleaning Services Does Divine Commercial Cleaning Offer for Medical Facilities?
Divine Commercial Cleaning provides specialised cleaning services tailored for a range of medical facility types, including dental clinics, aged care facilities, pathology labs and imaging centres, with local coverage across the Sunshine Coast and Brisbane. Services are customised to meet clinic-specific risk matrices, use TGA-listed disinfectants where required, and incorporate environmentally conscious methods and flexible contract arrangements without lock-in obligations. Divine emphasises a satisfaction guarantee and tailored solutions that align operational cleaning with facility infection prevention plans and audit requirements. For facility managers seeking compliance support, Divine offers quote requests and compliance assessments to map cleaning scope to NSQHS and Queensland Health expectations.
How Are Dental Clinics and Aged Care Facilities Cleaned Differently?
Dental clinics prioritise aerosol management, rapid operatory turnover and meticulous instrument-area cleaning; protocols focus on timed between-patient surface disinfection, suction and chair controls, and strict segregation of instrument processing zones. By contrast, aged care cleaning emphasises resident safety, frequent communal-area cleaning, outbreak management protocols and gentler product selection to reduce respiratory or skin sensitivities among residents. Both settings require documented procedures, staff training and audit evidence, but the emphasis shifts: dental settings require rapid, equipment-focused disinfection, while aged care balances infection control with resident wellbeing and frequent high-touch area cleaning. Tailored cleaning plans reflect these differing priorities while maintaining overall compliance.
Recognising these distinctions ensures that cleaning teams use appropriate products and schedules for each facility type to protect patients and occupants.
What Tailored Solutions Address Pathology Labs and Imaging Centres?
Pathology labs and imaging centres require cleaning solutions that protect sensitive equipment and avoid corrosive residues while maintaining disinfection standards; this means selecting non-corrosive disinfectants, coordinating cleaning windows with technical staff, and applying protocols that protect sampling integrity. Tailored solutions include scheduled deep cleans outside operating hours, use of equipment-safe wipe systems and strict separation of specimen handling areas from public zones. Coordination with facility biomedical or technical teams ensures that cleaning methods do not damage scanners or analysers and that disinfection aligns with operational safety. Documented processes and joint sign-off procedures provide assurance for both clinical staff and facility managers.
How Does Divine Commercial Cleaning Ensure Compliance and Quality in Medical Facility Cleaning?
Divine Commercial Cleaning ensures compliance through structured staff training, competency assessments, regular audits with digital documentation and a continuous improvement approach that aligns with NSQHS and Queensland Health expectations. Training modules focus on infection prevention, PPE use, colour-coded microfibre systems and correct application of TGA-listed disinfectants; competencies are validated by observed audits and refresher schedules. Audit programmes combine routine, terminal and ad-hoc outbreak-response checks, producing digital records, photographs and ATP results where appropriate to create an auditable trail for facility managers. Client reporting includes corrective action tracking and access to product MSDS on request, supporting transparent regulatory compliance.
What Training and Certifications Do Cleaning Staff Receive for Infection Prevention?
Cleaning staff receive infection-control-focused training that covers correct cleaning sequences, disinfectant dilution and contact times, PPE selection and colour-coded systems to prevent cross-contamination, with practical competency checks such as observed cleanings and shadowing. Training is refreshed on a scheduled basis and after incidents, and records are maintained to demonstrate ongoing competence for auditors. Emphasis on scenario-based drills (spill response, outbreak interventions) ensures staff can apply protocols under pressure and that knowledge translates into measurable performance. These measures create a workforce capable of delivering consistent, compliant cleaning in clinical environments.

Dianne, originally from Rockhampton, hails from a business-oriented family, with her father owning electrical stores and her uncle serving as Mayor. Moving to the Sunshine Coast at 13, she later pursued a rewarding real estate career and raised three children. As a single mom, she balanced university studies with domestic cleaning work. Armed with a Bachelor’s Degree in Business, majoring in Supply Chain Management, Dianne founded and grew Divine Commercial Cleaning into a thriving company. Her success is rooted in strong family mentorship, a positive attitude, and a solution-oriented approach, offering tailored cleaning services with integrity and strategic insight.

